Global Journal of Medical and Clinical Research Articles
Platelet Indices as Predictive Markers for Sepsis
Kofi Mensah1,2*, Kelvin M Maar3, Emmanuel B Borngreat3, Gabriel Abbam1, Samira Daud1, Rafiatu Salam3, Asana Alhassan3, Seth Kuntah4, Simon Bannison Bani3, Fatahiyatu Sayibu3, Augustine Badu5, Richard Duneeh6, Kpangkpari S Mwininyaabu4, Boniface Ukwah2, Felix Ejike Chukwurah2
1Department of Haematology, School of Allied Health Sciences, University for Development Studies, Tamale, Ghana
2Department of Medical Laboratory Science, Faculty of Health Science and Technology, Ebonyi State University, Abakaliki, Nigeria
3Department of Biomedical Laboratory Sciences, School of Allied Health Sciences, University for Development Studies, Tamale, Ghana
4Department of Family Medicine, Tamale Teaching Hospital, Tamale, Ghana
5Department of Medical Laboratory Science, Faculty of Allied Health Sciences, Koforidua Technical University, Koforidua, Ghana
6Department of Medical Laboratory Sciences, School of Allied Health Sciences, University of Health and Allied Health Sciences, Ghana
Cite this as
Mensah K, Maar KM, Borngreat EB, Abbam G, Daud S, Salam R, et al. Platelet Indices as Predictive Markers for Sepsis. Glob J Medical Clin Case Rep. 2025:12(1):022-028. Available from: 10.17352/2455-5282.000194Copyright License
© 2025 Mensah K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Sepsis continues to represent a significant global health challenge which is defined as an aberrant host reaction to infection that results in potentially fatal organ damage. Although recent strides have been made to improve early detection, prompt diagnosis still remains a big challenge. This study assessed the predictability of Platelet Distribution Width (PDW), Platelet count (PLT), and Mean Platelet Volume (MPV) in sepsis.
Methodology: This case-control study was conducted from June to September 2024 at the Intensive Care Unit of Tamale Teaching Hospital in Tamale, Ghana. Clinical records and Full Blood Counts (FBC) of 300 participants; including 150 participants diagnosed with sepsis and 150 participants without sepsis were assessed. The sample size of 300 participants was chosen to reflect equal representation between the two groups while maintaining statistical power for meaningful comparison. The smaller sample size, however, constrains subgroup analyses and wider generalizability. Further research with bigger sample sizes is recommended to confirm these results and explore other subgroups. Data was analysed using SPSS version 27.0, and statistical significance was set at p < 0.05.
Results: platelet count was found to be significantly lower (p = 0.001) in participants with sepsis (79.0[56.50 - 111.00] x 109g /L) than participants without sepsis (98.50[76.00 - 123.75]). PDW was significantly higher in participants with sepsis (12.10[8.45-14.35] fL) than their non sepsis counterparts (9.70[7.9 - 14.2] fL). Also, PCT was significantly higher in sepsis patients (0.21[0.14 - 0.32] %) than their counterparts without sepsis (0.19[0.13 - 0.30] %). PLT significantly predicted sepsis (AUC: 0.919, sensitivity: 72.8%, specificity: 96.6%, p < 0.001) in sepsis patients compared to MPV, PDW, and PCT.
Conclusion: Sepsis was significantly predicted by platelet parameters, with PLT being the most powerful predictor.
Introduction
Sepsis is an adverse immunological response to an infectious agent that precipitates a cascade of pathophysiological anomalies, ultimately culminating in significant organ dysfunction, low blood pressure, septic shock, and mortality [1].
The worldwide impact and frequency of sepsis represent significant challenges to public health; projections suggest that the condition accounted for 48.9 million new cases and 19.8% of all global deaths in 2017 [2]. Up to one-third of newborn fatalities worldwide are caused by sepsis. The World Health Organization, recognises newborn sepsis as a significant worldwide problem, with the highest prevalence in lower to medium-earning countries (LMICs) [3]. Although data are scarce in low-resource environments, such as sub-Saharan Africa (sSA), certain studies have found significant mortality rates [4]. Ghana’s newborn mortality rate is comparatively high across the country, with the Central Region having one of the worst rates at roughly 36 fatalities per 1000 live births [5].
Platelets are essential for immunological response, especially in sepsis and infection. Beyond clot formation, they also modulate inflammation, interact directly with pathogens, and affect the course of disease.
During sepsis, platelets interact with immunological mediators and pathogens to become active. They exhibit a range of receptors that enable direct attachment to bacteria by detecting Damage-Associated Molecular Patterns (DAMPs) and Pathogen-Associated Molecular Patterns (PAMPs). Platelet activation and aggregation may result from this interaction, which helps keep infections under control. However, Disseminated Intravascular Coagulation (DIC), which exacerbates organ dysfunction in sepsis, can be caused by excessive activation [6].
Thrombocytopenia has been reported as a common incidence of sepsis. The mechanisms leading to decreased platelet counts include reduced production, increased consumption, and heightened destruction [7]. However, limited studies have reported on platelets and their parameters as a predictive biomarker of sepsis, especially in Ghana. The study sought to add to the body of knowledge in the field and to emphasize the need to utilize platelet parameters, together with other clinical findings, in the determination of sepsis in resource-poor settings to influence prompt and timely therapeutic interventions to enhance patient outcomes.
Materials and methods
Study site & design
This case-control study was conducted at the Tamale Teaching Hospital, Tamale, in the Northern region of Ghana. TTH is the third largest hospital in Ghana which is situated on the main Tamale-Yendi Road in the eastern part of the city [8]. The facility is projected to have a land area of 646.90180 square metres and serves a population of approximately 233,252 people. The Tamale Metropolis is located between latitudes 9°16 and 9°34 North and longitudes 0°36 and 0°57 West on a geographical scale. Digitally, the hospital may be reached at NT-0101-5777 [8].
Study population and sample size
The study recruited 150 patients diagnosed with sepsis and admitted to the ICU. Also, 150 patients without sepsis who visited the haematology unit were selected as controls. Sample size calculations were as follows Using Kelsey’s formula for case-control studies.
, and
Ncases − Kelsey represents the number of Sepsis patients
“r” in this study, “r” stands for the ratio of the Sepsis group to the group without sepsis, which is 1:1; r = 1.
: denotes the critical value of the normal distribution at level α/2, and the critical value is 1.96 at a 95% confidence level where α is 0.05.
Zβ: denotes the normal distribution’s critical value at (this research employs an 80% power, β is 0.2 and the critical value is 0.84).
P1: prevalence rate of sepsis in Ghana, 29%.
P2: represents the prevalence of sepsis in the control group 50%
p1-p2: is the smallest proportional difference that is clinically meaningful and significant
The study consists of a sample size of approximately 300 Participants.
Inclusion and exclusion criteria
Patients diagnosed with sepsis and admitted to the ICU who consented to participate were recruited for this study. Patients without sepsis who visited the haematology unit were selected as controls. Pregnant women and patients with a history of cancer, liver disease, and blood disorders were excluded.
Diagnostic criteria
The Sepsis-3 standards redefine septic shock as a complication of sepsis characterized by persistent low blood pressure requiring vasopressors to maintain a Mean Arterial Pressure (MAP) of ≥ 65 mmHg and a serum lactate level > 2 mmol/L [9]. Sepsis-3 transitioned the definition of sepsis from a widespread inflammatory response to a dysregulated host response to infection causing organ dysfunction and life-threatening complications with a two-point increase in the SOFA score from the baseline.
Laboratory investigations
A whole blood volume of 5 ml was collected from each participant and later dispensed into tubes containing Ethylenediamine Tetraacetic Acid (EDTA) anticoagulant and carefully mixed. Analysis of whole blood indices was performed on the participants’ samples. A fully automated URIT 5380 haematology analyser was used to estimate haematological parameters at the Haematology Laboratory unit of TTH.
Ethical clearance
Ethical clearance was obtained from the Ethics and Review Committee of the University of Development Studies (UDS) (UDS/ RB/114 /24) and permission was obtained from the Research and Development unit of the Tamale Teaching Hospital before the commencement of the study.
Data analysis
Data entries were done with Microsoft Excel and analysed using a Statistical analysis tool (SPSS) version 27. The data was presented in tables and figures. Kolmogorov-Smirnov test and Shapiro-Wilk test were both used to determine the normality of the data set. Medians (25th - 75th percentiles) were used to present numerical data. Bivariate numerical data was compared using the Mann-Whitney U-Test. Correlations were performed by using logistic regression models. The sensitivity, specificity, and area under the curve (AUC) of the variables were analysed using ROC curves. Statistical significance was set at p < 0.05.
Results
Demographic characteristics of study participants
The socio-demographic profile of the participant pool included insights into their age, gender, and ethnicity (Table 1). Of the total participants, 187 (62.3%) were female while 113 (37.7%) were male, indicating a higher representation of females in the population. The average age of the participants was 31.35 yrs with a standard deviation of ± 23.4 yrs. Age groups were categorized as follows: 43 participants (14.3%) were neonates aged 0–30 days, 116 (38.7%) were infants aged 31–365 days, and 86 (28.7%) were toddlers aged 1–2 years. The largest group consisted of adults aged 19–64 years, accounting for 261 participants (8.7%). Participants aged 65–90 years constituted the smallest portion, accounting for 29 participants (9.7%) of the total sample. Ethnic distribution documented a total of 9 ethnic groups. The Dagomba ethnic group constituted the largest portion comprising 68 individuals (22.7%), followed by the Mamprusi with 55 participants (18.3%) and Ewe with 40 participants (13.3%). Other groups included Akan and Hausa, each with 32 participants (10.7%), Gonja with 28 participants (9.3%), Kusasi with 18 participants (6.0%), and Ga with 4 participants (1.3%).
Clinical characteristics of sepsis participants
Of the 300 total participants, 150 sepsis-diagnosed participants were admitted to the ICU over a period of time, during which clinical characteristics such as their length of ICU stay and source of infection were examined. The mean length of ICU stay was revealed to be [10.23 ± 6.2 days]. The majority of the participants (61.7%) had an ICU stay of less than 10 days, while 38.3% had an ICU stay longer than 10 days. Respiratory infections accounted for (76.5%), followed by bloodstream (12.8%), soft tissue (6%), and abdominal (4.7%) infections (Table 2).
Hemogram of the study participants stratified by the presence and absence of sepsis
Table 3 presents the haematological parameters of 300 study participants, categorised into those with sepsis (n = 150) and those without sepsis (n = 150). A lower platelet count was seen in sepsis patients [79.0(56.50 - 111.00) x 109g /L] compared to non-sepsis patients [98.50(76.00 - 123.75)], with a p-value of < 0.001. This indicates a statistically significant difference between the two groups. PDW was significantly higher in sepsis patients [12.10(8.45 - 14.35) fL] compared to non-sepsis patients [9.70(7.9 - 14.2) fL] with a p-value of < 0.001, indicating a statistical difference between both groups. PCT was significantly higher in sepsis patients [0.21(0.14 - 0.32) %] compared to non-sepsis patients [0.19(0.13 - 0.30)%] with a p-value = 0.025 showing a statistically significant difference between both groups. No statistically significant differences were observed for WBC count, WBC differentials, RBC count, HCT, and other indices, (Table 3).
Association between age, sex and sepsis severity
Table 4 presents the associations between age group, sex, and sepsis severity among the sepsis participants. Among male participants aged 1-10 years, the majority of cases were evenly split between sepsis and septic shock with each recording a frequency of 8 cases and a percentage of 44.4% while severe sepsis recorded 2 cases accounting for 11.1%. The 11-30 years age group had more incidence of sepsis cases than it did for severe sepsis and septic shock, recording 9 cases (50.0%), 3 cases (16.7%), and 6 cases (33.3%) for each respectively. 31-50 years had a slightly higher incidence of sepsis, recording 4 cases (40.4%) while severe sepsis and septic shock recorded 3 cases (30.0%) each respectively. 51-70 years recorded an equal number of incident cases of 2(50.0%) for sepsis and septic shock and no case of severe sepsis. 71-90 years recorded 1 case for sepsis (12.5%) 2 cases for severe sepsis (25.0%) and 5 cases for septic shock (62.5%). Among female patients, the highest burden of sepsis (9 cases, 33.3%) was observed in the 1–10 years age group, whereas severe sepsis was most prevalent in the 71–90 years group (4 cases, 25.0%). Septic shock in females was most common in the 11–30 years age group, accounting for 11 cases (39.3%), followed by 4 cases (25.0%) in the 71–90 years group. Severe sepsis in females demonstrated a wider distribution across all age groups. A p-value of 0.461, indicated no statistically significant difference in sepsis severity distribution across male age groups. Among female patients, the differences in sepsis severity across age groups were statistically significant, with a p-value < 0.001
Logistic regression analysis to evaluate the predictive power of platelet indices for diagnosing sepsis
An analysis was conducted to evaluate the predictive power of platelet indices for diagnosing sepsis. The results indicated that platelet count (PLT), Mean Platelet Volume (MPV), and Platelet Distribution Width (PDW) were significant predictors of sepsis. Specifically, PLT had a statistically significant negative association with sepsis diagnosis (B = -0.013, p < 0.001) and an odds ratio (Exp (B)) = 0.987, which means for every unit increase in PLT the odds of sepsis decrease by 1.3%, indicating that higher platelet counts were associated with a lower likelihood of sepsis. Conversely, MPV showed a positive association (B = 0.222, Exp (B) = 1.249, p < 0.001), indicating that, as MPV increases, the odds of sepsis increase by 24.9% suggesting that higher MPV values increase the likelihood of sepsis. Similarly, PDW was negatively associated with sepsis (B = -0.117, Exp(B) = 0.890, p = 0.009), showing that an increase in PDW decreases the likelihood of sepsis by 11%.
However, the Plateletcrit (PCT) did not significantly predict sepsis (B = -1.629, Exp (B) = 0.196, p = 0.153), implying that PCT’s relationship with sepsis could be due to random variation rather than a meaningful effect (Table 5).
ROC analysis to determine the predictability of PLT, MPV, PDW, PCT in sepsis
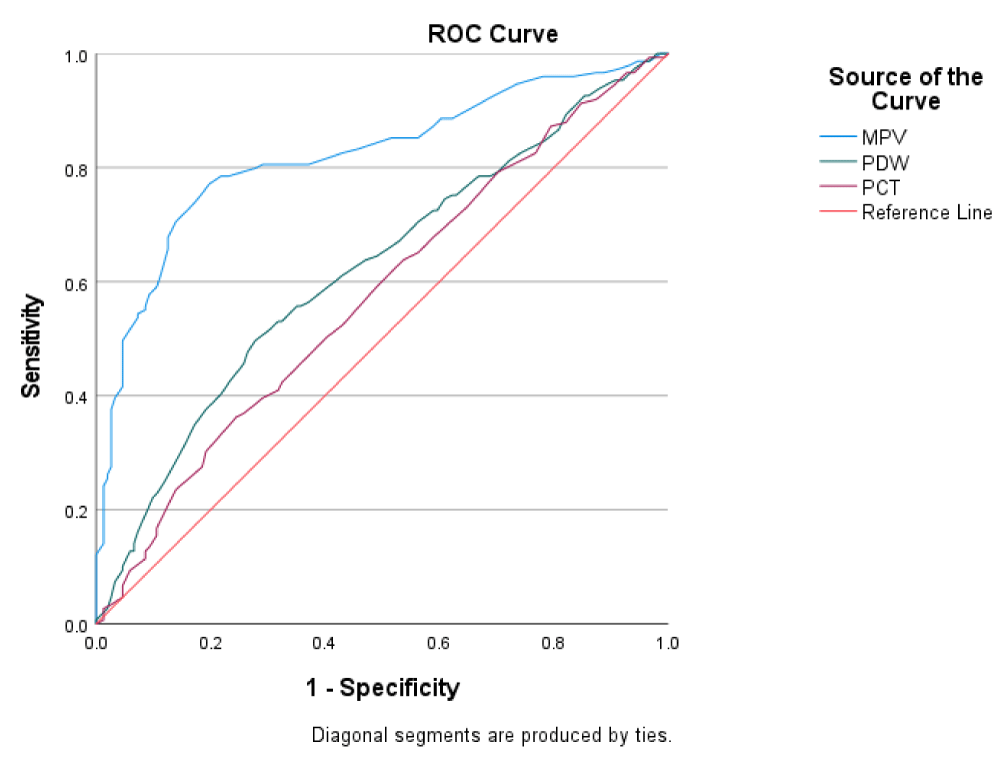
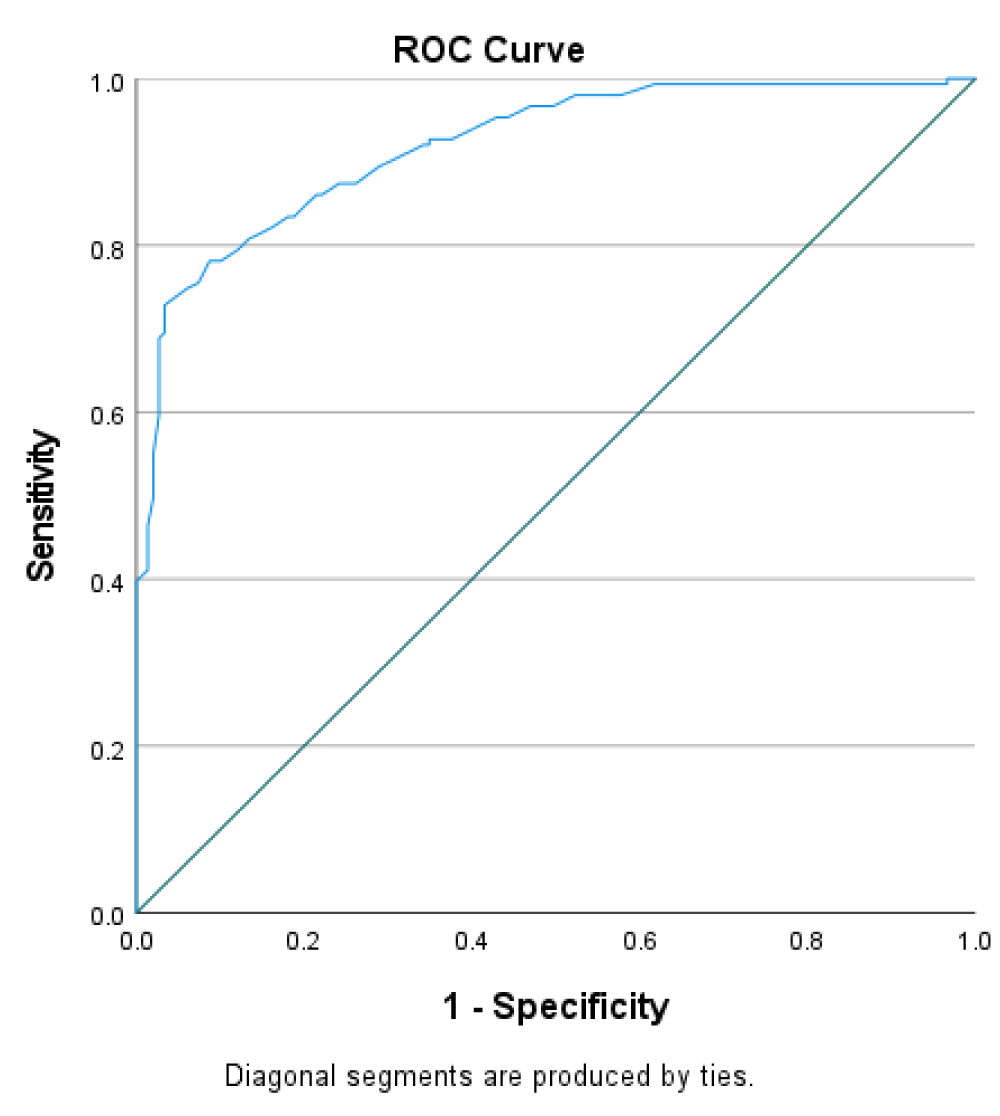
ROC was performed to assess the accuracy of platelet indices in sepsis prediction. The curves illustrate the trade-off between sensitivity and specificity for different cutoff values of the different platelet parameters. The area under the curve (AUC) of MPV was (0.821 (p < 0.001), indicating good discriminatory power, at the optimal cutoff point of 11.05, the test achieved a sensitivity of 77.2% and a specificity of 80.1%. The AUC for PDW was (0.621 p < 0.001) with a sensitivity of 72.5% and a specificity of 40.4%. PCT had an AUC of (0.575 p < 0.001) with a sensitivity of 95.3% and specificity of 9.3% (Figure 1). PLT also achieved an AUC of 0.919 with a sensitivity of 72.8% and a specificity of 96.6% (Figure 2).
Discussion
Sepsis is a substantial burden on global health and occurs due to a systemic inflammatory response to infection. The most vulnerable populations severely affected by sepsis are the elderly and young children [10]. Sepsis, unlike other conditions, has a range of clinical manifestations and complex pathophysiology, making prompt diagnosis difficult [11].
Patients with sepsis exhibited significantly lower platelet counts (79.0 x 109g/L) compared to negative controls (98.50 x 109g/L), with a p-value of < 0.001. Additionally, Platelet Distribution Width (PDW) was higher in sepsis patients (12.10 fL) than in controls (9.70 fL). A cohort of 500 adult patients was found to show thrombocytopaenia in sepsis cases in a study conducted by Beca, et al. [12], with platelet counts significantly lower in septic patients than in non-septic controls. An average platelet count of (85 x 109g/L) in a study conducted by Angele, et al. [13], supports the current study’s findings. A retrospective cohort study by Zhang, et al. [14] elaborated that lower platelet counts (less than 150 × 10⁹/L) were linked to increased mortality rates in patients with sepsis and septic shock. The similarities in findings are a result of platelet activation and consumption which are common occurrences in sepsis.
The length of stay of sepsis patients in the Intensive Care Unit (ICU) noted a majority of the participants had an ICU stay shorter than 10 days. This suggests that a shorter length of stay may be associated with improved patient outcomes while a longer stay is reflective of more severe illness. This aligns with Wang, et al. [15] in a different study, found that patients with declining platelet counts following intensive care unit admission were significantly more likely to die within 28 days (odds ratio of 2.464) than those with stable or rising counts after analysing data from over 22,000 septic patients. A previous study analysing data from 1,849 sepsis patients found that earlier admission to the ICU was associated with lower 28-day mortality rates and a longer length of stay was associated with higher mortality rates [16].
The current study also highlighted respiratory infections as the most common cause of sepsis. Ceccato & Torres [17], found similar findings in a study that highlighted that Community-Acquired Pneumonia (CAP) is the most common cause of sepsis, with 40% - 50% of sepsis patients presenting respiratory sources of infection. According to a paper by the Cleveland Clinic, respiratory infections, such as pneumonia, are common causes of sepsis, although infections can come from a variety of body areas [18].
A significant association between age group gender and sepsis severity was identified among female participants (p < 0.001), whereas no such association was observed among their male counterparts (p = 0.46). Angele, et al. [13] found no significant association in a study with a mixed age sample, although age was identified as a risk factor for increased severity. According to a different study, there were substantial disparities in fatality rates among sepsis patients by age group, but not by gender overall. For example, geriatric ladies were more likely to die than their male counterparts [19]. A retrospective analysis involving 265 sepsis patients showed that a higher proportion of males were affected (60.8%) however, gender did not significantly impact mortality rates. Deceased patients were older on average (mean age 69 vs. 43 years), suggesting that age is a critical factor influencing sepsis severity and outcomes [20]. A previous study found that the risk of death increases significantly with age, especially for males. In contrast, females showed a relatively stable risk of death until around age 80, indicating a different pattern of mortality related to age between genders. This implies that gender may have varying effects on outcomes depending on the age group.
The observed differences in age and sex associations may stem from variations in study populations and demographic characteristics. For instance, the current study’s significant association found in females may be explained by the study’s focus on a younger population, as hormonal and developmental factors in younger individuals could influence immune responses and sepsis outcomes differently.
In relation to the Predictive Power of Platelet Indices, Logistic regression analysis indicated that platelet count (PLT), Mean Platelet Volume (MPV), and PDW were significant predictors of sepsis. A study on platelet indices in critically ill septic patients linked increased mortality rates to aberrant platelet indices. PLT and MPV were found to be useful prognostic indicators in sepsis, and certain odds ratios for mortality were computed using these indices [21]. Higher MPV values are associated with increased severity and death, as established by Vélez-Páez, et al. [22], and PLT is a valid sepsis biomarker. A recent study suggested that elevated PDW levels correlate negatively with disease severity and can be used as an adjuvant predictor for sepsis [23]. This differs from findings in the current study which observed that an increase in PDW decreases sepsis. The observed dissimilarities could be due to the small sample size which led to bias and produced findings that deviated from trends observed in larger studies. Variations in laboratory equipment may have accounted for the observed difference.
Recommendation
Platelet indices demonstrate enormous potential as predictive markers for sepsis. Incorporating these indices as part of routine laboratory investigations for patients at possible risk for sepsis could aid early identification and timely intervention. Future studies involving larger sample sizes are needed to validate these findings across a broader population. Incorporating inflammatory biomarkers such as procalcitonin or c-reactive protein may provide a more robust diagnostic model.
Conclusion
Study participants with sepsis exhibited lower platelet counts and increased platelet indices (PDW and PCT). These findings were particularly notable in female participants, where a correlation was observed between age and severity of sepsis. In contrast, no such correlation was found among male participants. This suggests potential age- and gender-specific variations in how platelet indices and sepsis severity interact, warranting further investigation. Sepsis is significantly predicted by PLT, MPV, and PDW, with PLT being the most powerful predictor. It is recommended that Platelet indices should be combined with other biomarkers and clinical factors to enhance early detection and management of sepsis especially in resource-limited areas where other investigative panels are a challenge.
Conceptualization: Kofi Mensah, Kelvin M. Maar.
Study design: Kofi Mensah, Kelvin M. Maar, Emmanuel B. Borngreat.
Data curation: Kofi Mensah, Gabriel Abbam, Charles Nkansah, Felix Ejike Chukwurah.
Laboratory investigations: Kofi Mensah, Seth Kuntah, Samira Daud, Augustine Badu, Kpangkpari S. Mwininyaabu.
Statistical analysis: Kofi Mensah, Gabriel Abbam, Richard Duneeh, Rafiatu Salam.
Resources: Kofi Mensah, Kelvin M. Maar, Emmanuel B. Borngreat.
Supervision: Kofi Mensah, Simon Bannison Bani, Seth Kuntah, Boniface Ukwah.
Writing - original draft: Kofi Mensah, Kelvin M. Maar.
Writing – review & editing: Fatahiyatu Sayibu, Asana Alhassan, Seth Kuntah, Richard Duneeh, Felix Ejike Chukwurah, Augustine Badu, Simon Bannison Bani, Samira Daud, Boniface Ukwah, Kelvin M. Maar, Emmanuel B. Borngreat.
Data availability
All relevant data are available in this article and its supplementary file.
- Mangalesh S, Dudani S, Malik A. Platelet indices and their kinetics predict mortality in patients of sepsis. Indian J Hematol Blood Transfus. 2021;37(4):600. Available from: https://doi.org/10.1007/S12288-021-01411-2
- Fleischmann-Struzek C, Rudd K. Challenges of assessing the burden of sepsis. Med Klin Intensivmed Notfmed. 2023;118(2):68–74. Available from: https://doi.org/10.1007/S00063-023-01088-7
- Popescu CR, Cavanagh MM, Tembo B, Chiume M, Lufesi N, Goldfarb DM, et al. Neonatal sepsis in low-income countries: epidemiology, diagnosis and prevention. Expert Rev Anti Infect Ther. 2020;18(5):443–452. Available from: https://doi.org/10.1080/14787210.2020.1732818
- Lewis JM, Feasey NA, Rylance J. Aetiology and outcomes of sepsis in adults in sub-Saharan Africa: a systematic review and meta-analysis. Crit Care. 2019;23(1):1–11. Available from: https://doi.org/10.1186/S13054-019-2501-Y
- Adatara P, Afaya A, Salia SM, Afaya RA, Konlan KD, Agyabeng-Fandoh E, et al. Risk factors associated with neonatal sepsis: a case study at a specialist hospital in Ghana. Sci World J. 2019. Available from: https://doi.org/10.1155/2019/9369051
- Shannon O. The role of platelets in sepsis. Res Pract Thromb Haemost. 2021;5(1):27–37. Available from: https://doi.org/10.1002/RTH2.12465
- Dewitte A, Lepreux S, Villeneuve J, Rigothier C, Combe C, Ouattara A, et al. Blood platelets and sepsis pathophysiology: a new therapeutic prospect in critically ill patients? Ann Intensive Care. 2017;7(1):1–18. Available from: https://doi.org/10.1186/S13613-017-0337-7
- Kanton JF, Gyepi-Garbrah AP, Mensah ON, Richardson D, Kpikpitse D, Acquah H, et al. Knowledge and practices of home caregivers on neonatal danger signs pre-admission to Tamale Teaching Hospital, Ghana: an explorative descriptive study. BMC Pediatr. 2023;23(1):1–10. Available from: https://doi.org/10.1186/S12887-023-03879-5
- Singer M, Deutschman CS, Seymour C, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801. Available from: https://doi.org/10.1001/JAMA.2016.0287
- Jawad I, Lukšić I, Rafnsson SB. Assessing available information on the burden of sepsis: global estimates of incidence, prevalence and mortality. J Glob Health. 2012;2(1):10404. Available from: http://dx.doi.org/10.7189/jogh.02.010404
- Abu-Humaidan AHA, Ahmad FM, Al-Binni MA, Bani Hani A, Abu Abeeleh M. Characteristics of adult sepsis patients in the intensive care units in a tertiary hospital in Jordan: an observational study. Crit Care Res Pract. 2021. Available from: https://doi.org/10.1155/2021/2741271
- Beca J, Mcsharry B, Erickson S, Yung M, Schibler A, Slater A, et al. Hypothermia for traumatic brain injury in children - A Phase II randomized controlled trial. Crit Care Med. 2015;43(7):1458–1466. Available from: https://doi.org/10.1097/CCM.0000000000000947
- Angele MK, Pratschke S, Hubbard WJ, Chaudry IH. Gender differences in sepsis: cardiovascular and immunological aspects. Virulence. 2014;5(1):12. Available from: https://doi.org/10.4161/VIRU.26982
- Zhang R, Huang H, Lu S, Chen J, Pi D, Dang H, et al. Relationship between thrombocytopenia and prognosis in children with septic shock: a retrospective cohort study. Platelets. 2024;35(1). Available from: https://doi.org/10.1080/09537104.2024.2363242
- Wang K, Lu D, Wang F. Subphenotypes of platelet count trajectories in sepsis from multi-center ICU data. Sci Rep. 2024;14(1):1–12. Available from: https://doi.org/10.1038/s41598-024-71186-9
- Shibata J, Osawa I, Fukuchi K, Goto T. The association between time from emergency department visit to ICU admission and mortality in patients with sepsis. Crit Care Explor. 2023;5(5):E0915. Available from: https://doi.org/10.1097/CCE.0000000000000915
- Ceccato A, Torres A. Sepsis and community-acquired pneumonia. Ann Res Hosp. 2018;2(7):7–7. Available from: https://doi.org/10.21037/ARH.2018.06.01
- Sun B, Lei M, Zhang J, Kang H, Liu H, Zhou F. Acute lung injury caused by sepsis: how does it happen? Front Med. 2023;10:1289194. Available from: https://doi.org/10.3389/FMED.2023.1289194
- Ko RE, Kang D, Cho J, Na SJ, Chung CR, Lim SY, et al. Influence of gender on age-associated in-hospital mortality in patients with sepsis and septic shock: a prospective nationwide multicenter cohort study. Crit Care. 2023;27(1). Available from: https://doi.org/10.1186/S13054-023-04515-5
- Chang Z, Lu J, Zhang Q, Wu H, Liang Z, Pan X, et al. Clinical biomarker profiles reveal gender differences and mortality factors in sepsis. Front Immunol. 2024;15:1413729. Available from: https://doi.org/10.3389/FIMMU.2024.1413729
- Taha RS, Afandy ME, Elbadawi AH, Abd El Ghafar MS. Platelet indices in critically ill septic patients as a predictor of mortality. Egypt J Anaesth. 2023;39(1):56–62. Available from: https://doi.org/10.1080/11101849.2023.2168853
- Vélez-Páez JL, Legua P, Vélez-Páez P, Irigoyen E, Andrade H, Jara A, et al. Mean platelet volume and mean platelet volume to platelet count ratio as predictors of severity and mortality in sepsis. PLoS One. 2022;17(1). Available from: https://doi.org/10.1371/JOURNAL.PONE.0262356
- Ligi D, Della Franca C, Notarte KI, Goldrich N, Kavteladze D, Henry BM, et al. Platelet distribution width (PDW) as a significant correlate of COVID-19 infection severity and mortality. Clin Chem Lab Med. 2024;62(3):385–395. Available from: https://doi.org/10.1515/CCLM-2023-0625